Medical Center for Clinical Research

Medical Center for Clinical Research
6719 Alvarado Rd Suite #204
San Diego, CA 92120
Our Hours
Mon-Thur: 8:00AM to 5PM
Friday: 8:00AM to 2:30PM
Study Hotline
+1-619-344-0609
Main Office
+1-619-521-2830
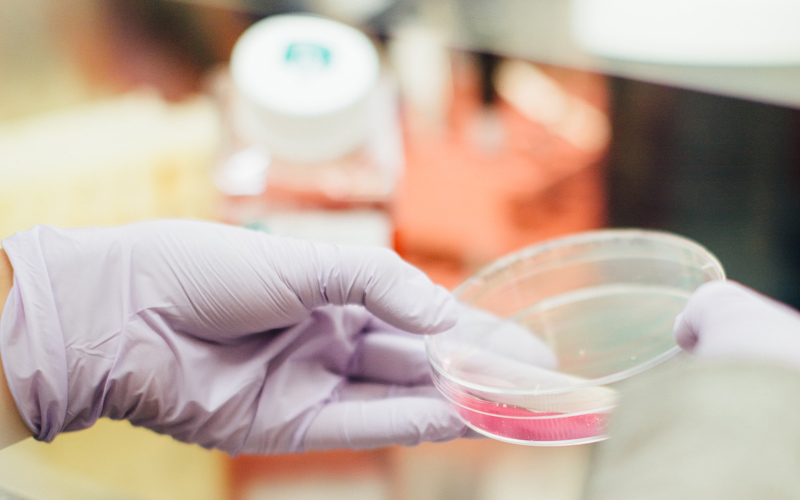
What we do
Medical Center For Clinical Research is a multi-disciplinary clinical research site that brings many years of experience and leadership in the research community to the San Diego area. Our research site effectively combines strategic accelerated volunteer recruitment and retention with high-quality clinical trial conduct practices. Our approach is uncompromising – each study conducted at our site is carefully planned and executed according to protocol and regulations with superior quality.
We conduct clinical research trials to advance health.
There are lots of reasons to participate in a medical study.
Browse and apply for a study today!